 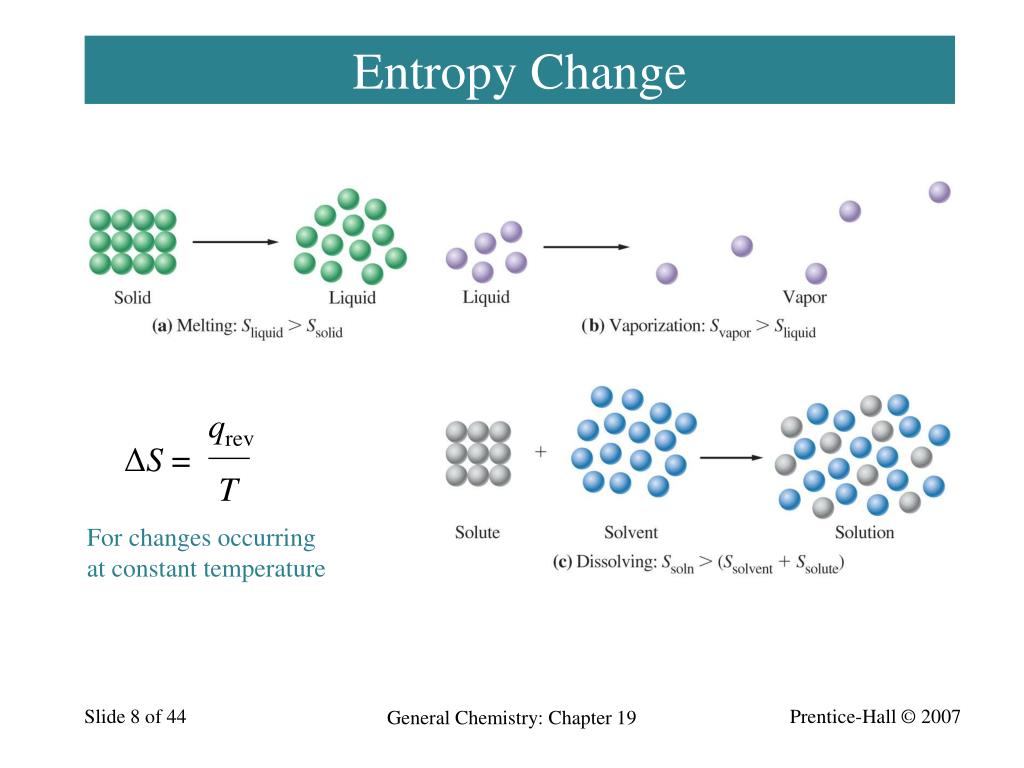
When entropy increases, a certain amount of energy becomes permanently unavailable to do work. Entropy is associated with the unavailability of energy to do work. In the second case, entropy is greater and less work is produced. The same heat transfer into two perfect engines produces different work outputs, because the entropy change differs in the two cases. All chemical reactions, from dissolving the. There is 933 J less work from the same heat transfer in the second process. All spontaneous processes increase the entropy of the universe the driving force of the universe is statistical. We noted that for a Carnot cycle, and hence for any reversible processes, We can see how entropy is defined by recalling our discussion of the Carnot engine. That unavailable energy is of interest in thermodynamics, because the field of thermodynamics arose from efforts to convert heat to work. 
Although all forms of energy are interconvertible, and all can be used to do work, it is not always possible, even in principle, to convert the entire available energy into work. Entropy is a measure of how much energy is not available to do work. Recall that the simple definition of energy is the ability to do work. Wind, water evaporation, gasoline, candy bar, water behind dam, cloud.ġ6.Making Connections: Entropy, Energy, and Work Which of the following type(s) of inhibitor can bind to the active site of an enzyme? A. the digestion of protein from food into amino acids. the formation of cellulose from individual glucose molecules, D. most particles have an amount of energy close to the average), we say that the entropy increases. one particle has all the energy in the universe and the rest have none) to a more probable distribution (e.g. A spontaneous reaction may involve an increase or decrease in enthalpy, it may involve an increase or decrease in entropy, but it will always involve a decrease in free energy that is a negative G. the replication of DNA from free nucleotides, C. When the way the energy is distributed changes from a less probable distribution (e.g. Entropy & the Second Law of Thermodynamics A reaction that is spontaneous. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system. Calculate the change in entropy of the reaction for the following chemical reaction at 298 K. entropy, the measure of a system's thermal energy per unit temperature that is unavailable for doing useful work. the synthesis of a phospholipid from glycerol and fatty acids, B. Which of the following chemical reactions would most certainly result in an increase in entropy of the system. Which of the following reactions is most likely to be exergonic? A. Reactions in which there is a negative change in free energy (ΔG) are: spontaneous and endergonic, B. Which reaction results in the greatest increase in entropy?Įntropy is often increased by: heat, B. always result in increased disorder of the systemħ. electron configuration and ionic chargeĬ. Which of the following statements explains why the melting of ice is a spontaneous. The two factors that determine whether a reaction is spontaneous or nonspontaneous areĬ. Which reaction results in the greatest increase in entropy A->2B. Melting is accompanied by an increase of energyġ0. Melting is accompanied by a decrease of energyĭ. Melting is accompanied by an increase of entropyĬ. Melting is accompanied by a decrease of entropyī. Which of the following statements explains why the melting of ice is a spontaneous reaction at room temperature and pressure?Ī. In which of these systems is the entropy decreasing?ĩ. Which reaction results in the greatest increase in entropy?Ĩ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
